The DISCSEEL® Procedure is the only minimally-invasive, patented procedure that treats back and neck pain resulting from torn, damaged spinal discs.
The DISCSEEL Procedure is proven to seal and heal damaged, torn spinal discs confirmed in placebo controlled studies.
Does Back Pain or Neck Pain Limit YOU?
You may have a spinal disc problem, such as a torn or damaged disc. Torn spinal discs can progress into degenerative disc disease, bulging or herniated discs, sciatica, or numbness in the legs and feet.
Consider the DISCSEEL Procedure.
The DISCSEEL Procedure utilizes a natural, Fibrin sealant, that is precisely injected into torn spinal discs to seal, heal and regrow disc tissue.
The DISCSEEL Procedure restores the health of your spinal discs and results in long-lasting pain relief.
Get to know the DISCSEEL® Procedure
Natural, Injectable Biologic Treatment
Seals and Heals Disc Tears
30-minute, Outpatient Procedure
Greater than 82% Success Rate
The DISCSEEL Procedure is a Flexible Option:
You can have the DISCSEEL Procedure even if you have previously had spine surgery or regenerative procedures (stem cell or PRP) that have failed.
Quick Facts
The DISCSEEL Procedure
Lasting Treatment for Chronic Back Pain – Seals, heals and promotes regrowth in torn and leaking spinal discs.
Minimally Invasive Procedure –Short 30-minute Outpatient Procedure.

Seals and Heals Torn Discs
Precise, Safe and Effective – Fibrin, an FDA-approved biologic, is injected into spinal disc tears to seal and heal.
Quick Recovery Time – no major downtime up and moving around sooner.
A specialty-trained, licensed and certified DISCSEEL Procedure physician can help you find long-lasting back pain relief.
DISCSEEL Procedure physicians are specially trained and certified to offer the procedure in their practices. To be offered this opportunity, interested physicians go through an application process and are required to complete, a methodical, hands-on, comprehensive training program.
Explore our growing list of physicians that are specially trained to offer the DISCSEEL® Procedure.










Find A DISCSEEL Procedure Physician near you.
Physicians interested in adding The DISCSEEL® Procedure to their practice INQUIRE HERE
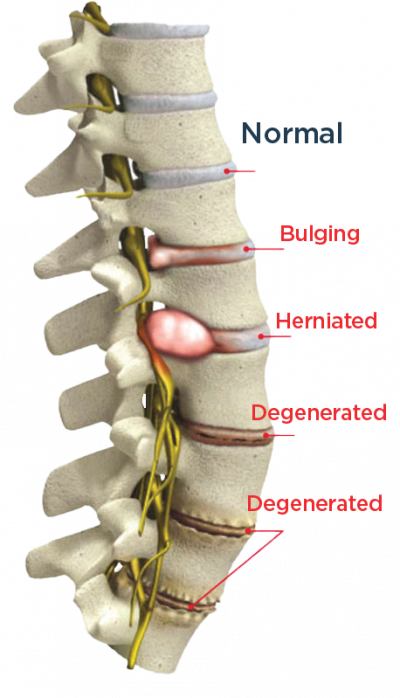
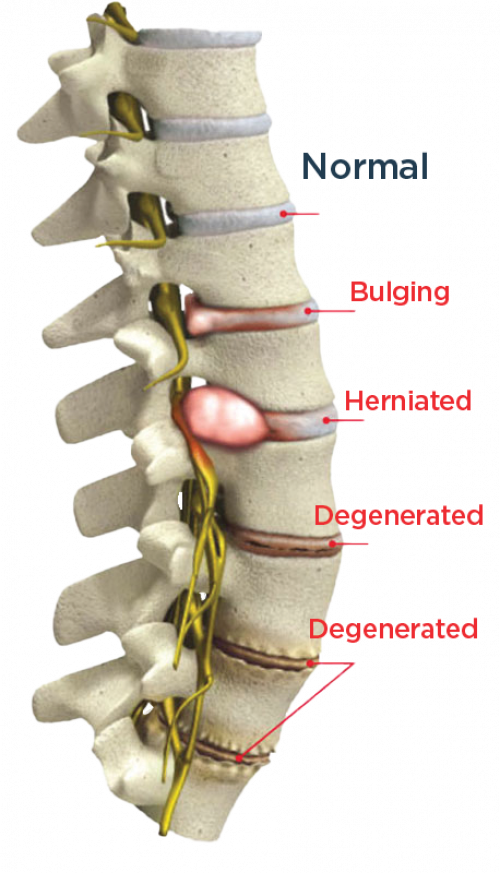
The DISCSEEL Procedure treats these conditions
- Annular Tears
- Bulging Discs
- Herniated Disc
- Degenerated Disc Disease
Most common cause of back pain is torn spinal discs (annular tears) that progress to bulging, herniated or ruptured discs that leak their inner fluid on to surrounding spinal nerves causing pain and inflammation.
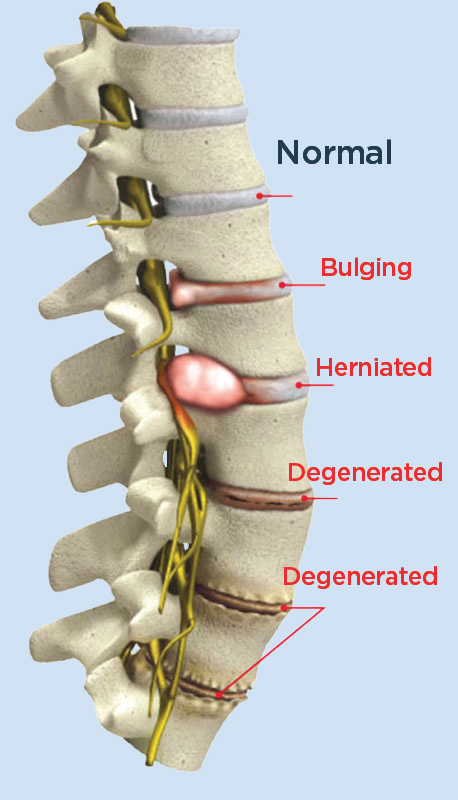
See how leaky spinal discs cause pain and inflammation. (WATCH VIDEO 0:27)
What is Leaky Disc Syndrome?
Leaking discs are the most common cause of pain and symptoms in the lower back, leg, neck, and arm.
Find A DISCSEEL Procedure Physician near you.
Patient Testimonials
Watch these personal stories of real patients’ experiences with the DISCSEEL Procedure and the difference it has made in their lives.
Click the Video to Watch
Rick had intense back pain for ten years and tried multiple treatments that failed. The DISCSEEL Procedure solved his pain. Rick says now he can focus on his business instead of his back pain.
Click the Video to Watch
Erica now says that the DISCSEEL® Procedure has “100% turned her life around” and allowed her to get back to the things she loves, like running.
Click the Video to Watch
Sean wanted to find a solution to his back pain that didn’t involve putting hardware in his spine, so he chose the DISCSEEL® Procedure. Now he’s living pain-free and has been able to return to the lifestyle he loves.